Scientists Build a New Womb to Uncover Why Babies Struggle Early
In the quiet hum of a lab, a team of scientists has put something incredible together. They built an artificial womb. It is not a place you see on a beach or a hat. It is a small, carefully arranged environment where life can grow.
Why did they do it? Every pregnancy starts with an embryo. But a lot of those embryos do not survive the first weeks. Roughly 60 % of them fail to attach to the uterus or disappear soon after. That causes many miscarriages. Scientists want to know why that happens.
Why the Mystery Matters
Mistakes in early pregnancy cause big problems. They can hurt a mother’s health. They can make future family planning harder. Doctors keep asking: What goes wrong inside? When an embryo sits on its way, something stops it.
Common guesses focus on the embryo itself. Some say the embryo might be weak. Others think the uterus might not welcome it. Or maybe the blood flow isn’t right. Scientists don’t have a simple answer yet.
Creating a Controlled Environment
Imagine opening a window to a garden. In the garden, you can see every flower and leaf. Scientists wanted a garden that looks like a womb. They gave the environment the right temperature. They turned the water into a liquid that is safe for a baby inside.
The key is keeping the gases balanced. Baby cells need oxygen. They need the right mix of carbon and nitrogen. The artificial womb does that in a closed circuit. It keeps the fluid from getting dirty. It stops bacteria from entering. Everything stays calm.
They also made the walls in the right shape. A real uterus is not flat. It curves. The artificial womb mirrors that shape. The baby feels like it is not alone. The walls help the embryo attach, just like in a real body.
What the Team Did
The scientists used a small portion of a real human uterus. But to keep it clean, they took tissues from healthy donors. They repurposed these strands in the tank.
They put them into a device that feeds the baby. The machine also provides the correct pressure. The baby holds the right amount of fluid.
To see what works, they grow several embryos in fewer machines. The growth is measured. The scientists look. They watch if the embryo sticks to the wall, if it grows a bit, and if it stays healthy.
Additionally, they look inside the tank at a microscopic level. They study how the baby talks to the walls. They watch how cells move and grow.
What Were the Results?
After weeks, some embryos grew happily. Others did not. The scientists noticed that the current problems happen when the embryo could not sit on the wall like in real life. They felt that something is missing in that early stage.
When they observed embryos that did attach, they had a bigger chance to survive. Those embryos grew a little more. They showed many healthy cells. Those are the points scientists want to keep for the next step.
Overall, the experiment shows that giving the embryo the right environment is very important. The artificial womb can handle most parts of an early pregnancy. The researchers can now test new ideas safely.
What This Means for the Future
Imagine a single woman who wants to have a baby but has problems attaching. She might use this artificial womb to help the embryo grow. It could reduce the number of miscarriages. The article shows that this idea is not only hope but a working practice at least in limited cases.
For people who are often, noisy in the first weeks, the artificial womb can isolate them from external pressure. The baby can grow in a calm, controlled way. That means it will have a better chance to keep developing.
Scientists are not ready to use this new womb with real babies yet. They need more tests. They also want to see if the babies stay healthy after leaving the artificial womb. However, the path forward looks promising.
Why This is Not Just a Lab Idea
Without solutions, people lose hope. A large number of miscarriages affect families. The artificial womb can bring comfort. It also brings an answer to the question: Why do babies not function at the earliest stages? We are moving forward.
What People Ask About the Solution
- Will the babies be safe? Scientists performed several safety checks. The environment keeps bacteria out. They also used minimal amounts of chemicals and checked them thoroughly.
- Can this help for other conditions? For people with implantation issues, the womb can help. It creates a nice environment where the embryo can attach. That can reduce stress in the mother.
- Will the baby’s life be normal? Current data shows that the baby grows fine inside the artificial womb. Future research will confirm if the newborns stay healthy later.
- How can doctors use it? Doctors may look at it as a supplement, not a replacement. They can choose to use it when normal implantation fails. This is a new tool for complicated pregnancies.
Some Concerns and Ethics
Many people are worried about the idea. Some think that an artificial womb is a step that is too far. Others worry about the cost. They also wonder if it will change how humans think about parenthood.
Scientists are careful to listen. They do not want to push the technology on families before it is safe. They also ask for public input: Who should get it? In what cases? These are important questions as the technology develops.
What Are the Next Steps?
Scientists will run more experiments. They will let more embryos grow in a larger number of artificial wombs. They will take detailed notes on all stages. These notes help scientists craft the best guidelines.
They also want to combine the device with other medical technologies. For example, combining it with blood tests. This way doctors can see the embryo’s health in real time.
Every step helps reach the bigger aim: reduce the chance of miscarriage. Less men or women will have to endure the painful loss of a baby. The hope is that life can breathe in a safer space.
Little Steps, Big Impact
The response shows how a small invention can change a big problem. A small womb may help contain the right conditions for a baby. That is a small, bright idea in life. It could be the first answer to an age-old question: why do babies often fail early?
You Can Be Part of the Conversation
When scientists ask for feedback, they listen. If you are a parent, a doctor, or just a curious reader, you can use your voice. The conversation about artificial wombs is still fresh. Your thoughts help shape how the future will look. It will not just be in a lab. It will touch everyday life with babies.
So keep following the story. You will see new tests, new results, and a growing hope that this technology moves from the bench to the bedside. The world needs this kind of care to support families true to the chance of a new life. After those 60 % of embryos, there is hope for a better future.
Key Takeaway
Scientists built an artificial womb. They want to learn why many embryos fail early. The results show that a controlled environment helps the embryo attach and grow. This breakthrough could one day reduce miscarriage rates and bring more safety to early pregnancy. It is a small, yet powerful step toward healthier births.
Scientists Record Human Embryo Implanting In Real Time – A Breakthrough
For many years, doctors and scientists have wondered how a tiny fertilised egg attaches itself to a mother’s womb. That moment, called implantation, is key to a healthy pregnancy. Yet, until now, the picture was a mystery. Now, a team in Barcelona has captured it live, making history in the field of reproductive science.
Why This Matters
Every successful pregnancy starts with implantation. If it goes wrong, the embryo may not grow, or the pregnancy may fall apart. Knowing exactly what happens can help doctors spot problems early.
Patient stories show how scary it can be. A woman who had a twin pregnancy failed to carry it to term. She wondered why the process didn’t work. If doctors could see the embryo’s actions, they might offer better treatments.
Researchers also want to understand how different eggs perform. Not all embryos are the same. Some are strong, some weak. Knowing the difference can guide fertility clinics.
The Challenge Before Today
Until today, scientists could only look at static pictures. They had to rely on a limited number of still frames taken from ultrasound or biopsy. These snapshots missed the action between day five of a pregnancy and the first ultrasound, a gap that left many questions unanswered.
In a cumulative sense, the whole process had been a black box. There were no videos, no animations, no live demonstrations. Doctors could guess, but they could not see.
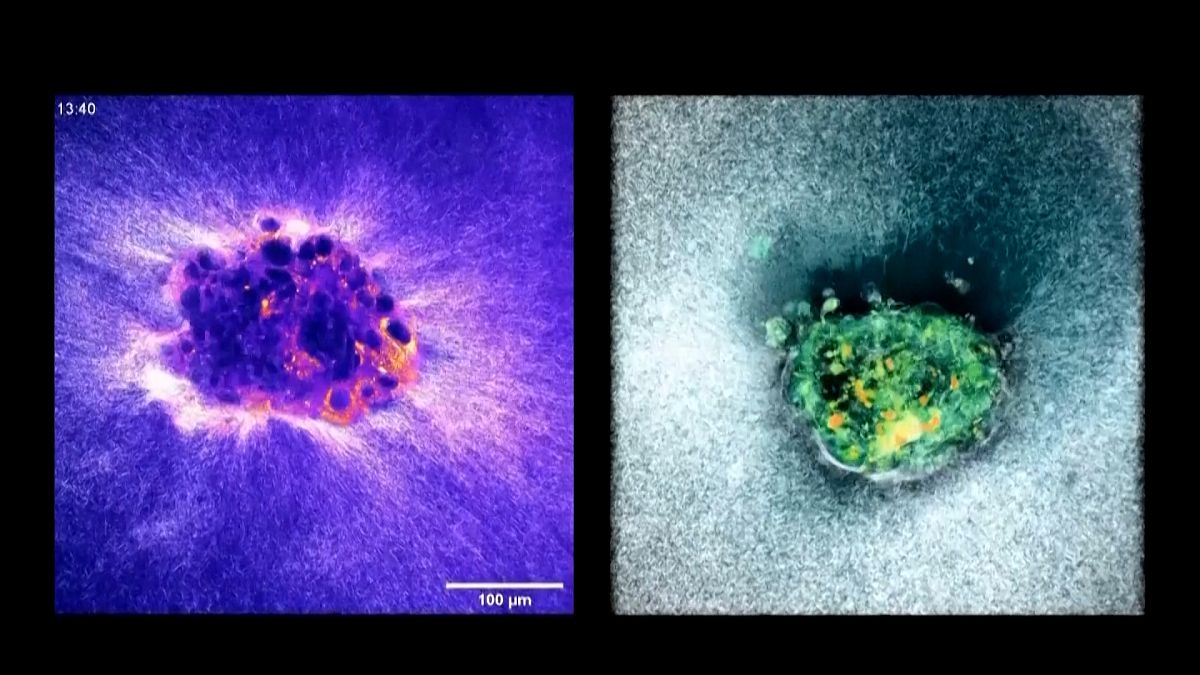
The New Image‑Makers
In Barcelona, a group at the Institute for Bioengineering of Catalonia, or IBEC, built something new. They created a model that looked just like the inside of a real womb. It was made using collagen and proteins that are found inside human body tissue.
By laying out this special surface, they gave embryos a place to try to stick. The embryos used in the study were donated for research and kept in a safe, ethical way.
Samuel Ojosnegros Martos, the head of this research group, explained that the embryos are free-floating. That means they are not attached to anything. By week 5, they must find and hook onto the womb lining, or they will be lost. Doctors can only see them weeks later. Until now, the steps between those days were unknown.
The Setup
- They built a miniature womb like the lining of a uterus.
- They used the real tissue’s material – collagen and proteins.
- They placed the embryos on top of this baseline.
- They filmed the entire process with high‑resolution cameras.
- They tracked how strong each embryo was.
The film shows the embryos digging and pulling themselves into the matrix. The motion is not random. The embryo digs its tiny fingers into the lining and leverages force to slide in.
What We Learned
Healthy embryos pulled strongly. Low‑quality embryos did not pull as hard. In simple terms, the force an egg exerts appears to be a marker for success.
Martos said, “We discovered that the human embryo is very invasive. It can dig a hole inside the matrix and pull itself very strongly.”
Of note, the team also used mouse embryos in a test. Those mice behaved differently. Instead of diving in, they stayed on top of the surface. That means human embryos act differently, and the mouse model does not match the real situation.
Implications for Fibroblast and Endothelial Studies
Scientists working on blood vessel formation and connective tissue found that a healthy embryo can potentially stimulate surrounding cells. The implanting process might recruit fibroblasts – cells that create structure—and endothelial cells – cells that form blood vessels. This coordinated activity is needed for the embryo to grow its own blood supply.
When the embryo fails to attach, no this neat signalling happens. In those cases, the pregnancy may fail early because there is no proper blood supply.
Significance for Clinical Practice
1. Quality Assessment— Clinics can now refine how they choose which embryos to transfer.
2. Early Interventions— If doctors watch a video of the implanting embryo, they might spot problems earlier. They could use drugs or therapy at a cheaper time.
3. Better Counseling— Couples can get a realistic picture of what the embryo is doing. The story becomes less mysterious.
What Comes Next?
Scientists plan to replicate the setup in a human womb environment. That will allow them to mimic the exact temperature and hormone levels that naturally exist. They will tailor the matrix to match the real uterine lining more accurately.
These future tests will involve more embryos, giving them a statistically robust result. The team will also test how changing the material composition affects the embryo’s pulling strength.
Another front is genomics. Scientists will look at genes that allow the embryo to be invasive. They will compare high‑quality and low‑quality embryos to identify the genetic markers that drive implantation.
Why it Feels Human
Throughout the study, the researchers held the living embryos in hands. The footage shows movement that feels alive. The film turns a cold scientific problem into a gentle story about human life.
For one mother-in‑waiting, the video was a relief. She said, “Seeing the embryo move was like a window into my child’s life.” This emotional connection will help the public understand very well what science is all about.
In Short
You will go back to the womb to check the progress of an implanting embryo, thanks to this new technology. Future research might be less hidden. Doctors will know exactly when the embryo is doing its best. Then, they can help people have babies with less worries.
Key Takeaways
- We have captured an embryo, for the first time, planting itself in a womb‑like surface in real time.
- Healthy embryos pull more strongly, implying quality directly affects implantation.
- Mouse embryos behave differently, which means human models cannot rely on mice for this purpose.
- This discovery opens a new path for better fertility treatments, and for early diagnosis of implantation failures.
- Researchers plan to refine the model and investigate genetics to understand the mechanisms behind the implantation process.
Every step in this research shines a fresh light on the human body. It underlines that even in the tiniest stages, humans are capable of creating a beautiful, complex life from something so small.
Why it matters
Why Some Embryos Don’t Stick
Impressing a baby into the womb is a tough job.
Even though 60 % of embryos are either not settled or vanish soon after, that’s the main reason people lose pregnancies early.
Scientists think that learning why and how embryos get stuck can make IVF and clinics much better.
What the Research Says
Dr Tim Child, a reproductive‑medicine professor at Oxford, says this paper is “fascinating.”
It digs into the mystery of good embryos that mismatch the uterus.
The science is hard to sell, but if we get the answers, results could rise.
Why Genetics Isn’t the Whole Story
- Embryos that carry the right set of chromosomes have an 80 % chance of settling.
- But they still fail 20 % of the time.
- The lab‑to‑uterus transfer is the most common failure point.
- Future medicine might give women a little nudge to help the embryo attach.
What This Means for IVF
When doctors check a fertilized embryo carefully, they can spot the good ones.
But the window for the embryo to touch the lining of the uterus is still small.
Clinical teams could now look at how to improve that window – maybe with new drugs.
That could lift the success rate dramatically.
How We Can Hope for More Successful Starts
With each new study, we get closer to knowing what the embryo likes and needs.
For people hoping to conceive or doing IVF, better understanding means more chances to grow that tiny life safely.
Bringing the science from a lab to the clinic is the next step.
And if this research sticks, it’s a big win for families worldwide.